Site author Richard Steane
The BioTopics website gives access to interactive resource material, developed to support the
learning and teaching of Biology at a variety of levels.
|
Site author Richard Steane
|
The BioTopics website gives access to interactive resource material, developed to support the
learning and teaching of Biology at a variety of levels.
|
|
Oxidation |
Oxidative |
OP takes place alongside electron transport chain in |
| Combination with oxygen | Phosphorylation is a process - addition of phosphate |
inner membrane of |
| Removal of hydrogen | Part of aerobic respiration |
mitochondria inside |
| Loss of electrons (clue: OIL RIG) |
Other parts of respiration which provide reactants ... glycolysis, link reaction & Krebs/TCA/citric acid cycle |
cells of |
| Opposite of oxidation is ... reduction |
Eukaryotes (animals, plants, fungi +) |
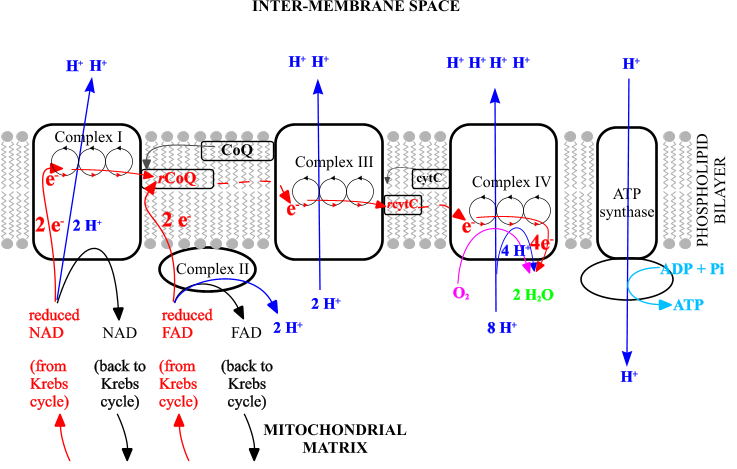
| Reduced NAD (Nicotinamide adenine dinucleotide) |
H+ (protons) and electrons (e-) from Reduced NAD/FAD enter Electron Transport Chain |
ADP (Adenosine diphosphate) |
|
| Sometimes written as NADH, or NADH/H+ [also reduced FAD flavin adenine dinucleotide FADH - similar but less energy] |
Electrons are passed along the ETC & stay in the membrane: 4 subunits (complexes I-IV) |
H+ are pumped/ deposited at the other side of the membrane- in the inter-membrane space |
Pi is inorganic phosphate [not P alone] Adenosine is adenine + ribose - as in RNA |
| A coenzyme - absorbs energy from breakdown of molecules in other reactions |
Electrons pass from compound to compound in ETC (redox reactions) losing energy at each stage |
H+ builds up so there is a concentration gradient between inside and outside of mitochondrion - chemiosmosis |
This energy from H+ moving down its concentration gradient is used to phosphorylate ADP (combine ADP and Pi) |
| Provide reducing power: H+ & e- (proton and electron donation) |
Electrons' energy is used to pump H+ across the membrane |
H+ passes though ATP synthase back into centre of mitochondrion |
ADP +Pi (+energy) ATP [energy is not a reactant] ADP +Pi |
| And are oxidised in the process |
Electrons and protons are passed to oxygen: final electron/proton acceptor 4H+ + 4e- + O2 |
Oxidative |
|
| and recycled ģ RH2 + NAD+ NADH + H+ + R |
The reason why we breathe in |
ATP (adenosine triphosphate) is the energy currency of cells ATP + H2O ΔG░ = -30.5 kJ/mol |
|